① About Prostate cancer
Prostate cancer is one of the most common malignancies among men worldwide, with approximately 1.6 million new diagnoses and 366,000 deaths reported annually. The prostate gland is typically located inferior to the bladder, anterior to the rectum, and encircles the urethra. It is a small, walnut-shaped gland in the male body that produces seminal fluid, which nourishes and transports sperm. Genetic alterations in prostate epithelial cells drive the initiation and progression of prostate cancer, and without timely intervention, the disease can result in substantial morbidity and mortality. In the United States, approximately 11% of men will be diagnosed with prostate cancer during their lifetime, with incidence increasing markedly with age; the annual numbers of newly diagnosed cases and deaths are approximately 268,490 and 34,500, respectively. Therefore, early detection is a critical determinant for improving patient prognosis.
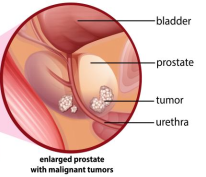
② About Prostate Cancer Susceptibility Genes
The most important known risk factors for prostate cancer are age and inherited genetic variants. According to relevant studies, more aggressive forms of prostate cancer—such as early-onset disease, tumors with metastatic lesions, and cancers with higher Gleason scores—are more likely to harbor known hereditary cancer susceptibility and driver genes. In the presence of certain pathogenic variants in cancer risk genes, early detection and diagnosis of prostate cancer are critical for controlling disease progression.
③ Project Introduction
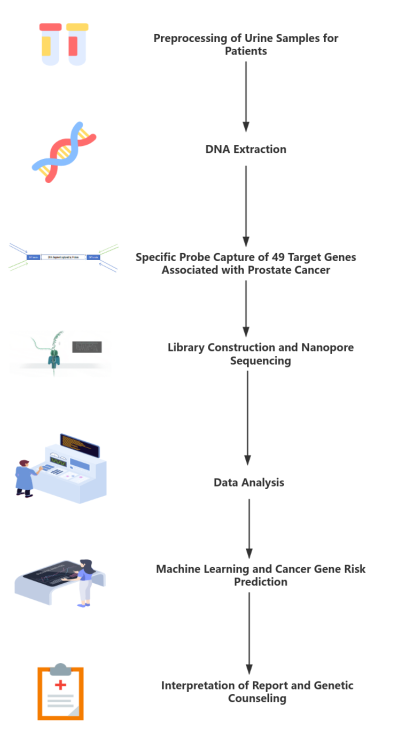
Based on the nanopore sequencing platform combined with a probe-based target enrichment approach, a total of 49 prostate cancer–related genes are captured and sequenced to comprehensively screen for susceptibility and driver genes associated with prostate cancer. In addition, disease subtype–specific probes are designed to enable the detection of prostate cancer–related gene fusions. By integrating machine learning algorithms with multiple curated databases, prostate cancer–associated risk scores can be predicted. Compared with conventional testing methods and second-generation mNGS technologies, this approach enables ultra-long-read sequencing and real-time data analysis, streamlines the overall workflow, improves detection efficiency, reduces labor costs, and maximizes data utilization. Collectively, it meets clinical demands for early screening and diagnosis of prostate cancer susceptibility and driver genes.
④ Project Process
⑤ Prostate Cancer-related Gene
Inherited genes (N=13)-遗传性基因 |
ATM BRCA1 BRCA2 BRIP1 CHEK2 EPCAM MLH1 MSH2 MSH6 PALB2 PMS2 RAD51C RAD51D等 |
Prostate tumor-specific genes (N=18)-特异性基因 |
ATM BRAF CDKN1B CHD1 CTNNB1 FOXA1 HRAS KDM6A MED12 MYC NKX3.1 PDE4B PDE10A PIK3CA PTEN等 |
Genes involved in fusions (N=18)-融合驱动基因 |
ACSL3 C15orf21 CANT1 DDX5 EST14 FOXP1 HERV-K HERVK17 HNRPA2B1 KLK2 SLC45A3 TMPRSS2 ERG ETV1 ETV4等 |
The above only lists some of the genes
⑥ Project Advantage
Comprehensive target coverage: A panel of 49 prostate cancer–related target genes was selected based on multiple authoritative databases and expert consensus. The panel encompasses hereditary genes associated with germline mutations, tumor-specific genes related to somatic mutations, and special fusion driver genes. Specific probes were designed accordingly, enabling more comprehensive detection coverage.
Combined detection modalities: The assay supports simultaneous DNA and RNA co-detection and also allows joint sequencing and integrative analysis of familial samples within the same run.
High data accuracy: Built on a third-generation nanopore sequencing platform, the approach leverages ultra-long-read sequencing and real-time data analysis, enabling more accurate detection of gene-associated variants.
High turnaround efficiency: From sample collection through processing, sequencing, and data analysis to report generation, the entire workflow can be completed within 7–15 days.
⑦ Target Population
Male patients aged ≥50 years presenting with one or more symptoms suggestive of prostate cancer.
Male individuals aged 40–45 years and older with a family history of prostate cancer.
Other testing indications: male individuals carrying pathogenic variants in genes such as BRCA1 or BRCA2.
High-risk male populations with elevated PSA levels and abnormal findings on digital rectal examination (DRE).