① Detection background
In the process of assisted reproductive technology, the incidence of aneuploidy in embryos of advanced age women and some infertile groups is relatively high, often leading to embryo arrest, miscarriage or implantation failure, while traditional morphological assessment is difficult to effectively identify abnormal embryos, so it is necessary to introduce more accurate detection methods. PGT-A (Preimplantation Genetic Testing for Aneuploidy) can detect all 23 pairs of chromosomes before embryo transfer, and select embryos with normal chromosomes. Its main purpose is to reduce the risk of recurrent implantation failure, miscarriage and birth defects caused by the transfer of abnormal embryos, so as to improve clinical outcomes of assisted reproductive technology.
② Project Introduction
Nanopore sequencing has the advantages of ultra-long read length, no GC preference, and can accurately resolve highly repetitive and other complex regions, with comprehensive technical advantages. Non-invasive pre-embryonic aneuploidy testing (niPGT-A) developed based on this platform is different from traditional PGT-A in detection methods. It is through the analysis of cfDNA in the culture medium of blastocysts, combined with bioinformatics analysis, to comprehensively assess the chromosome number before embryo transfer, thus avoiding the potential damage to embryo cells caused by taking trophoblast cells, reducing the impact on embryo development, achieving the selection of preferred embryos, and providing clinical transplantation decision-making references, which helps to improve the success rate of IVF, clinical pregnancy rate and embryo utilization rate.

③ Detection content
Detection name | Non-invasive pre-embryonic aneuploidy testing (niPGT-A) |
Detection content | 1. Screen for non-heritable aneuploidy of all 23 pairs of chromosomes
2. Can detect ≥4Mb chromosome deletion or duplication
3. Chimera abnormalities >10Mb and >30% |
Inspection cycle | 7-10 days |
Target audience | 1. Advanced maternal age (age 38 or above)
2. Unexplained recurrent spontaneous abortion (two or more recurrent spontaneous abortions)
3. Unexplained recurrent implantation failure (three or more embryo transfers or four to six high-grade cleavage-stage embryos or three or more high-grade blastocysts transfers all fail)
4. Severe teratozoospermia |
Detecting sample types and preservation methods | Collect the corresponding culture medium in the blastocyst culture dish with a pipette gun from the blastocyst (embryo development to stage 4 or above). After the pipette gun head is blown and sucked in the droplet three times, collect all the culture medium and transfer it into a labeled collection tube, and send it for inspection as soon as possible; if inspection cannot be performed as soon as possible, the collected samples need to be placed in a -20℃ refrigerator. Logistics transfer requires the use of dry ice to send samples for inspection. |
④ Detection process
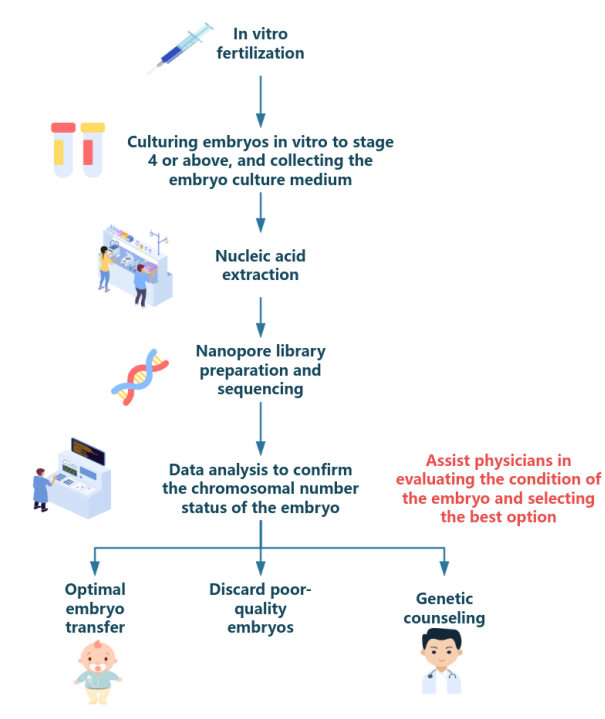
⑤ Detection advantage
l The procedure is entirely non-invasive, ensuring enhanced safety throughout the operation. It requires only blastocyst culture medium, thereby circumventing potential damage associated with traditional embryo biopsy and providing an additional safeguard for embryonic development. Reports are generated rapidly, significantly minimizing the duration of embryo exposure.
l Leveraging the real-time reading capabilities of nanopore sequencing, combined with our in-house developed high-efficiency analytical pipeline, redundant data are precisely filtered, substantially compressing the overall testing workflow.
l The long-read advantage of the nanopore platform allows for the traversal of complex chromosomal regions, yielding higher detection accuracy. Moreover, the integration of machine learning algorithms enables in-depth analysis of signal and sequence features, ensuring the utmost precision of results.
l The testing cost is economically favorable, as reusable nanopore chips markedly reduce consumable expenses. High-accuracy, single-pass identification of euploid embryos minimizes both the financial and psychological burdens associated with repeated embryo transfers.