Recently, the National Medical Products Administration announced the approval of the 1.2-class traditional Chinese medicine innovation drug, Houttuynia cordata (Akelestat) soft capsules, under the Shengnuoji's Shen Noki. This is also the first approved traditional Chinese medicine new drug in 2022, and the approval of this product for marketing has once again ignited the market's attention on traditional Chinese medicine for tumor diseases. Data from Menin Network shows that in recent years, the sales scale of traditional Chinese medicine for tumor diseases in China's public medical institutions has been about 20 billion yuan, with a year-on-year increase of 9.33% in the first half of 2021.
20 billion market stirs up another wave! The first anti-tumor traditional Chinese medicine 1st new drug of the year is approved, rushing to the 1 billion large variety
Recently, the National Medical Products Administration announced that it has conditionally approved the上市 of Shen诺基's 1.2-class traditional Chinese medicine innovation drug, Epimedium (A克拉定) soft capsules, through the priority review and approval procedure, providing new treatment options for patients with hepatocellular carcinoma.

The product submitted its marketing application in April 2021, which was accepted by the CDE. It was approved for marketing nine months later, becoming the first new traditional Chinese medicine to be approved for marketing in 2022. Records show that Shen诺基 is a wholly-owned subsidiary of Shen诺基. Previously, Shen诺基 calculated that the expected sales of the Epimedium tablet soft capsule in 2022 would achieve marketing, and the revenue of the product would reach 900 million-1 billion yuan in 2024, which could achieve the company's overall break-even.
Recently, East China Pharmaceutical Group announced that its wholly-owned subsidiary, Hangzhou Zhongmei East China Pharmaceutical, has signed a product exclusive market promotion agreement with Shengnuo Ji's wholly-owned subsidiary, obtaining the exclusive market promotion rights for the soft capsules of Epimedium in 27 provinces in mainland China.
It is worth mentioning that a total of 12 new traditional Chinese medicine drugs were approved for marketing in 2021. From the perspective of therapeutic indications, no products were approved for the treatment of oncological diseases.
The sales situation of traditional Chinese medicine in the terminal of public medical institutions in China with malignant tumors (Unit: billion yuan)
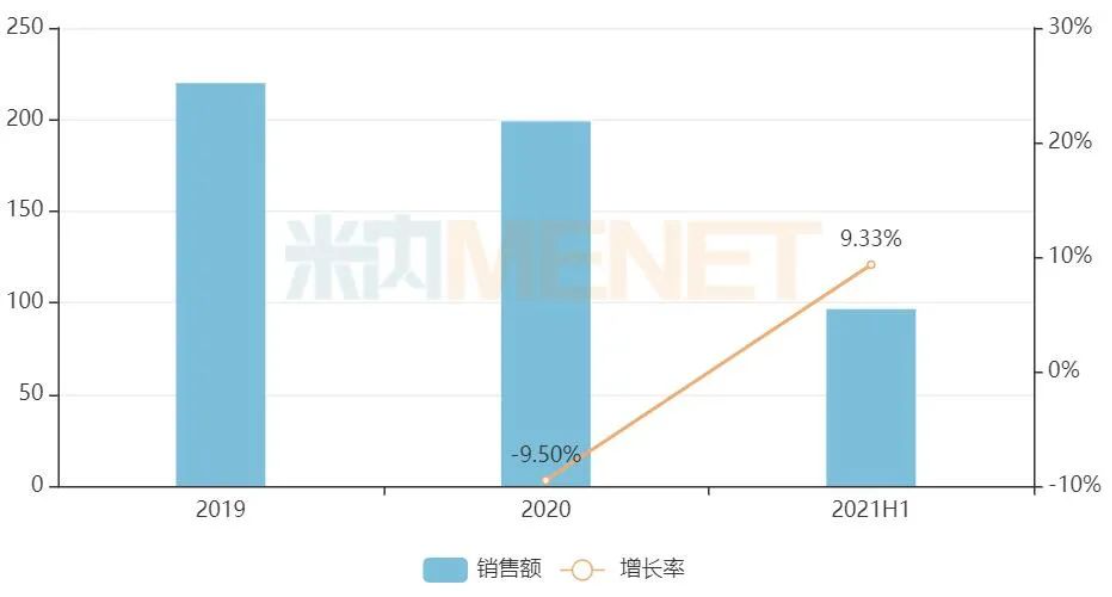
Source: Menetech China Public Medical Institutions Terminal Competition Landscape
Minew Data shows that in recent years, the terminal sales scale of traditional Chinese medicine for oncological diseases in China's urban public hospitals, county-level public hospitals, urban community centers, and township health centers (hereinafter referred to as China's public medical institutions) has been about 20 billion yuan, with a year-on-year increase of 9.33% in the first half of 2021. From the perspective of sales channels, urban public hospitals are still the main force, but the market share is on the decline; county-level public hospitals and urban community centers are steadily growing. From the top 10 manufacturers, Zhejiang Kanglait Pharmaceutical, Shaanxi Dongtai Pharmaceutical, and Guizhou Yihai Pharmaceutical are in the top three.
TOP10 ranking changes! Two major products rise over 40%, a new 10 billion brand or a new high
Top 10 Traditional Chinese Medicine Products for Oncological Diseases in Public Medical Institutions in China during the First Half of 2021
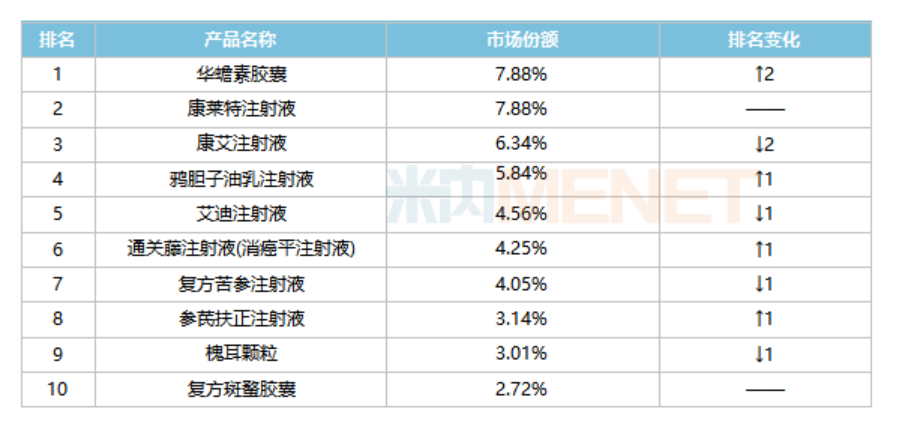
Source: Menetech China Public Medical Institutions Terminal Competition Landscape
The top four products among the top ten in sales all exceeded 500 million yuan, and six of them were exclusive products. Among them, Tongguan Teng Injection (Xiaoying Ping Injection) had the fastest growth rate, and Huacain Capsules, with a growth rate of more than 40%, ranked first.
Top 10 brands of traditional Chinese medicine for oncological diseases in public medical institutions in China in the first half of 2021.
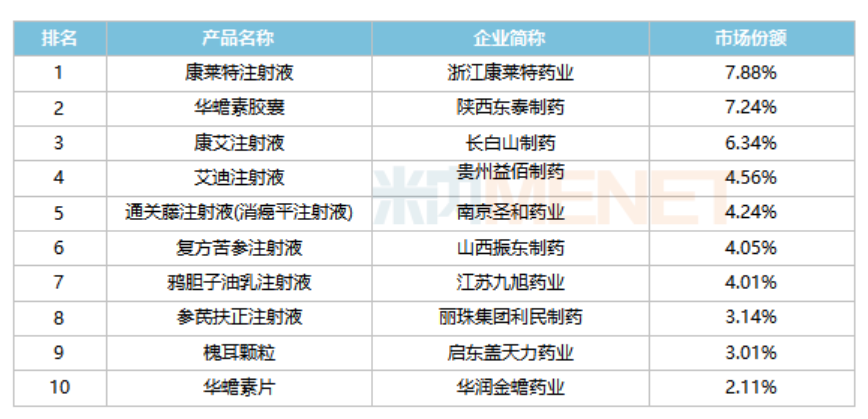
Source: Menetech China Public Medical Institutions Terminal Competition Landscape
Among the top 10 brands, Zhejiang Kanglait Pharmaceutical's Kanglait Injection has been in the lead for many years; Shaanxi Dongtai Pharmaceutical's Hua Chuan Su Capsules, after breaking the 1 billion yuan sales barrier in 2020, increased by nearly 40% in the first half of 2021, and is expected to set a new high for the whole year. In terms of growth rate, five brands, including Li Min Pharmaceutical's Shen Qi Fu Zheng Injection and Shanxi Zhendong Pharmaceutical's Compound Bitter Cnidium Injection, have all exceeded 20%.
Huacain Capsulesis mainly used for middle and late stage tumors, and can also be used for chronic hepatitis B and other diseases. It was included in the National Basic Drug List in 2018 and in the 2021 edition of the National Medical Insurance Directory as a Class A variety, limited to the use of cancer pain. Data from Minne Network shows that in recent years, the sales of Huacain Capsules in China's public medical institutions have exceeded 1 billion yuan, and the year-on-year growth in the first half of 2021 reached 41.37%. Currently, only 2 companies have production approval for this product, among which, Shaanxi Dongtai Pharmaceutical has the largest market share, with a year-on-year growth of nearly 40% in the first half of 2021, and it is expected to reach a new high in the whole year.
Chuanhengteng Injection (Xiaoxianping Injection)is a natural herbal medicine extracted from the effective parts of a single medicinal plant, Chuanhengteng, which contains effective anticancer components. It is used for the treatment of tumors such as esophageal cancer, gastric cancer, and liver cancer, as well as adjuvant therapy in combination with radiotherapy and chemotherapy. According to data from Minne Network, sales of Chuanhengteng Injection (Xiaoxianping Injection) in China's public medical institutions increased by more than 50% in the first half of 2021 compared to the same period last year, making it the fastest-growing product among the top 10. Currently, only two companies, Nanjing Shenghe Pharmaceutical and Tonghua Jinma Pharmaceutical Group, have production approval for this product, with Nanjing Shenghe Pharmaceutical having the largest market share.
Good policies are coming out frequently! 3 anti-tumor traditional Chinese medicine class I new drugs are ready to go!
The approval of new traditional Chinese medicine for clinical trials in malignant tumors in recent years
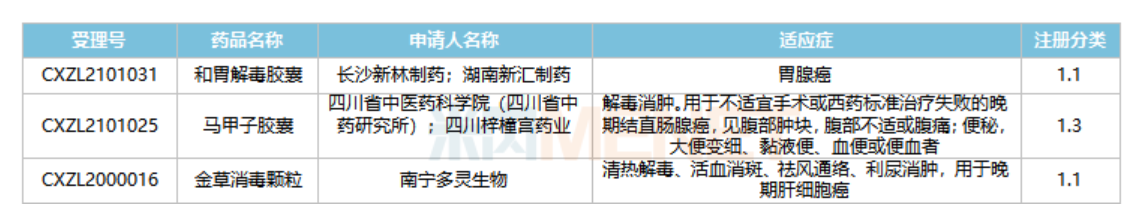
Source: CDE official website
Recently, the approval of the soft capsule of Epimedium for marketing has once again ignited the market's attention to traditional Chinese medicine in the treatment of tumor diseases. CDE data show that in recent years, two 1.1类 new drugs and one 1.3类 new drug have been approved for clinical trials. Among them, the indications of the胶囊are gastric adenocarcinoma; the indications of the capsule are advanced colorectal adenocarcinoma; the indications of the granules are advanced hepatocellular carcinoma.
At the beginning of 2022, the state issued a number of major policies in the field of traditional Chinese medicine.
On January 5th, the CDE issued a notice for public consultation on the "Technical Guidance for Communication and Exchange in the Context of the 'Three-Combined' Registration Review Evidence System for Traditional Chinese Medicine". The notice points out that in order to promote the construction of a registration review evidence system for traditional Chinese medicine that combines theoretical knowledge, clinical experience, and clinical trials, and to guide the submission of clinical professional communication and exchange applications for traditional Chinese medicine new drugs developed under the "Three-Combined" review evidence system.
On January 7th, the National Medical Products Administration issued three announcements, namely, "Technical Guidance Principles for the Revision of Safety Information Items in Instructions for Traditional Chinese Medicine already on the Market (Trial)", "Technical Guidance Principles for the Research of Samples for Toxicological Studies of New Traditional Chinese Medicine (Trial)", "Technical Guidance Principles for the Writing of Traditional Chinese Medicine Theoretical Supporting Materials for New Compounds (Trial)", and "Technical Guidance Principles for the Writing of Instructions for Ancient Classic Famous Traditional Chinese Medicine Compound Formulations (Trial)".
On January 10, the National Development and Reform Commission and other 21 departments issued the "14th Five-Year Plan for Public Services", which clearly states that in the field of medical and health services, efforts should be made to increase the training of grassroots traditional Chinese medicine personnel, encourage and guide grassroots medical and health institutions to provide appropriate traditional Chinese medicine services; adhere to the principle of equal importance and complementary advantages of traditional Chinese medicine and Western medicine, vigorously develop traditional Chinese medicine services, and give full play to the unique role of traditional Chinese medicine in disease prevention, treatment, and rehabilitation.
On January 15, the State Administration of Traditional Chinese Medicine and the Office of the Working Group for Promoting the Construction of the "Belt and Road" jointly issued the "Planning for the High-Quality Integration of Traditional Chinese Medicine into the Construction of the "Belt and Road" (2021-2025)", which clearly takes the inheritance and innovation of traditional Chinese medicine as the main line, aims to promote the modernization, industrialization and going to the world of traditional Chinese medicine, focuses on key regions, key countries and key international organizations, inherits the essence, adheres to the original innovation, fully plays the unique advantages and role of traditional Chinese medicine in preventing and treating diseases, and serves the people of the countries participating in the construction of the "Belt and Road". To contribute to the promotion of building a community of health for mankind.
With the continuous release of favorable policies for traditional Chinese medicine at the national level in recent years, the industry has formed a policy support from multiple dimensions such as market access, payment, and evaluation and approval, and the subsequent detailed implementation of policies in various places will promote the industry to enter a period of rapid development.
Source: Official website of the National Medical Products Administration, Menin database, company announcements.